 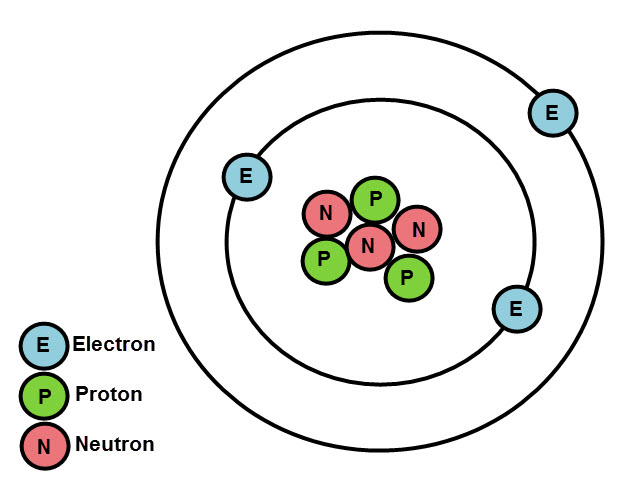
Postulates of Bohr’s model of an atom are:. To explain the stability of an atom, Neils Bohr, a Danish Physicist in 1913, proposed a new model of an atom. 
But this actually does not happen and the atom is quite stable. Thus, the orbit of the revolving electron will keep on becoming smaller and smaller, following a spiral path and ultimately the electron should fall into the nucleus, which means the atom should collapse. Whenever a charged particle-like electron is revolving around a central force like that of the nucleus, it losses energy continuously in the form of radiations. The electrons revolve around the nucleus in well-defined orbits.ĭrawbacks of Rutherford’s model of an atomĪccording to Rutherford’s model, an atom consists of a small heavy positively charged nucleus in the center and the electrons were revolving around it.The size of the nucleus is very small as compared to the total size of an atom.There is a positively charged center in an atom called the nucleus in which all the mass of an atom resides.On the basis of scattering experiments, Rutherford put forward a model of atom known as Rutherford’s Nuclear Model This small heavy positively charged body present within the atom was called a nucleus. Since the number of alpha particles that deflected back is extremely small, which shows that the heavy body present in an atom must be occupying a very small volume.Since some alpha particles were deflected back and we know that alpha particles are heavy particles, they could only be deflected back upon striking some heavy object inside the atom.The alpha particles which deflected through large angles were those which passed close to the positive body. The alpha particles which deflected through small angles were those which passed at a distance pretty far from this positive body. As some of the alpha particles were deflected by small angles, and since alpha particles are positively charged they could be deflected only by some other positively charged body present within the atom.Since most of the alpha particles passed through the foil without undergoing any deflection, there must be sufficient empty space within the atom.through an angle greater than 90 degrees.įrom these observations, Rutherford drew the following conclusions:. only one in 12000 were deflected back i.e. Some particles were deflected through small angles and a few were deflected through large angles.Most of the particles that are 99.9% of the particles passed through the foil without undergoing any deflection.Wherever alpha particles struck the screen a flash of light is produced at that point on the screen.įrom these experiments, Rutherford made the following observations:. He observed the scattering of the rays after hitting the foil by placing a circular zinc sulfide screen around the metal foil. These were obtained from radium placed in the cavity of block of lead and made into a fine beam with a slit. 
Rutherford’s scattering experiment: In 1911, Rutherford performed an experiment in which he bombarded a thin foil of a heavy metal like gold with a beam of fast-moving alpha particles which are doubly ionized helium ions or He2+ ions, each having 2 units of positive charge and 4 units of mass i.e. So, the atom as a whole is electrically neutral.Īlthough Thomson’s model explained that the atom is electrically neutral, the result of experiments carried out by other scientists could not be explained by this model.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
